

Higher and foundation tiers
Many of the physical properties of the alkanes depend on the size of the particular alkane molecule. These properties include: their boiling and melting points as well as viscosity. We’ll also look at how easily different alkane molecules ignite and burn, that is how flammable they are. Now the viscosity of a liquid is simply how runny or how easily it will flow and flammability is how readily a substance catches fire. These two particular properties are important for hydrocarbons since their main use is as fuels.
Let's start with looking at the trends and patterns in the melting and boiling points of the alkanes. The table below shows the melting and boiling points of the first 10 alkanes; can you spot any trends or patterns in the data?
| alkane | molecular formula | boiling point/°C | melting point/°C | state at room temperature |
|---|---|---|---|---|
| methane | CH4 | -161 | -182 | gas |
| ethane | C2H6 | -88 | -183 | gas |
| propane | C3H8 | -42 | -188 | gas |
| butane | C4H10 | -0.5 | -138 | gas |
| pentane | C5H12 | 36 | -130 | liquid |
| hexane | C6H14 | 69 | -95 | liquid |
| heptane | C7H16 | 98 | -90 | liquid |
| octane | C8H18 | 126 | -57 | liquid |
| nonane | C9H20 | 151 | -51 | liquid |
| decane | C10H22 | 174 | -30 | liquid |
The trends in the boiling point and melting points are easy to spot from the table; the larger the alkane molecule the higher the boiling and melting points. This trend is probably what you were expecting:
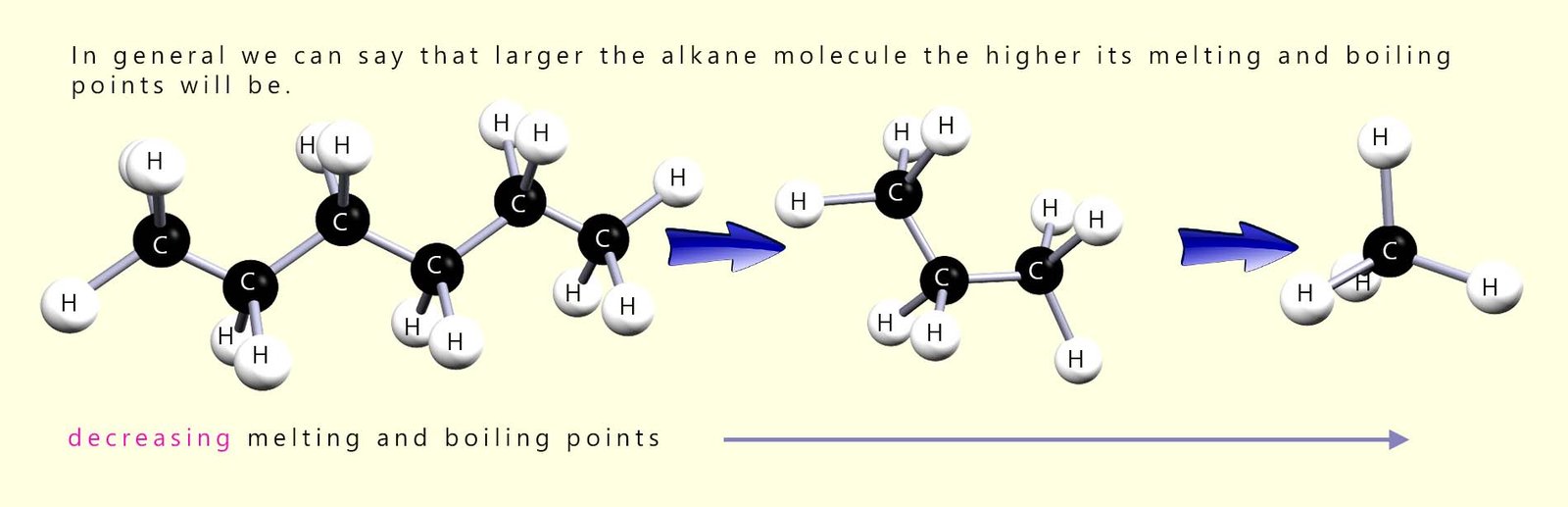
The larger the alkane molecule the larger its molecular mass will be and the more intermolecular bonding there is between different molecules, so its boiling and melting points both increase. This is outlined in the diagram below; where the two alkane molecules ethane and butane are shown. Butane being a larger molecule than ethane has more intermolecular bonding between different butane molecules. Stronger intermolecular forces make it harder for molecules to slide past each other, so the liquid becomes more viscous.
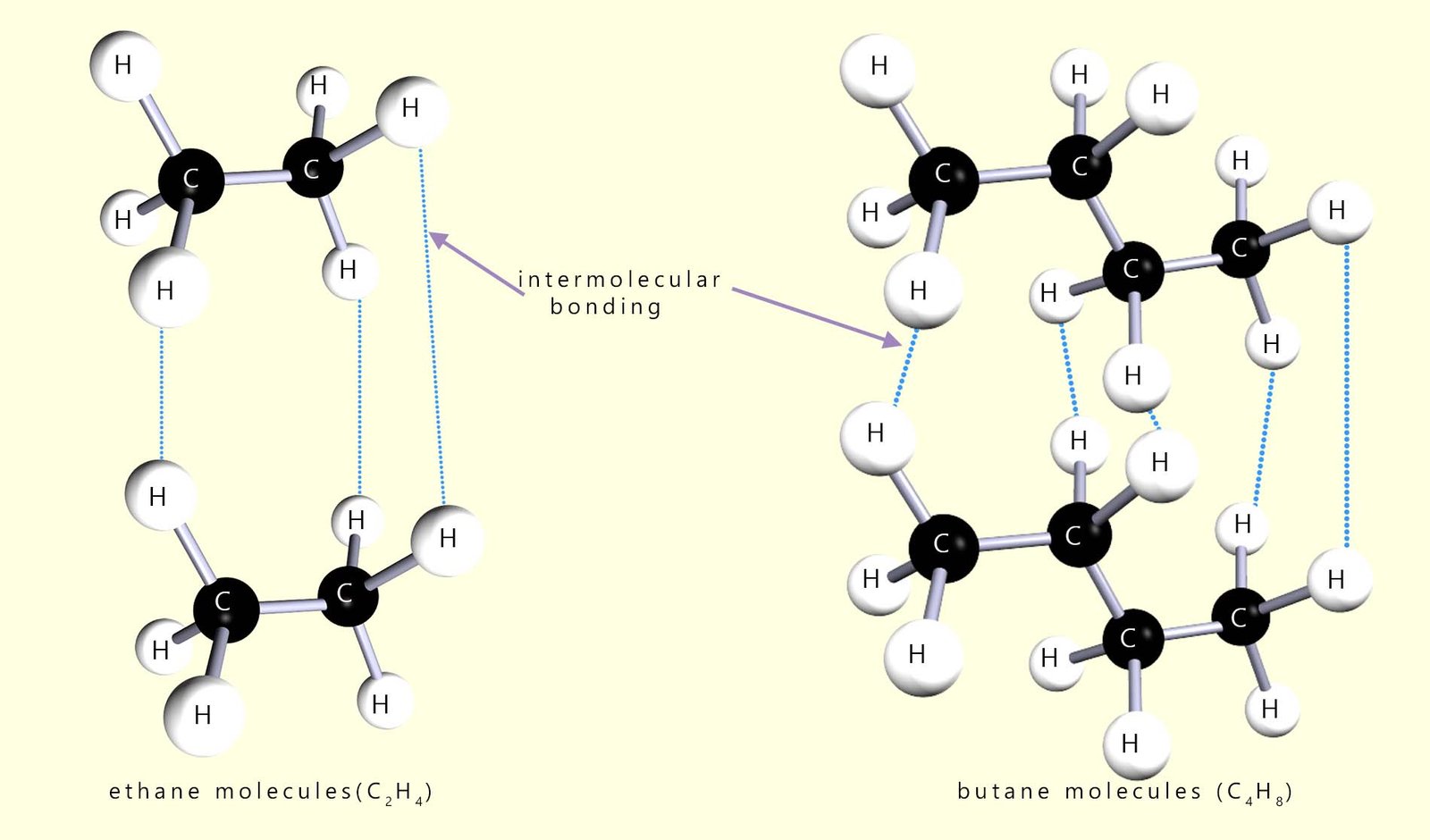
The general trend is similar for the melting points. The first 4 alkanes are all gases at room temperature while the larger alkane molecules from pentane (C5H12) to hexadecane (C16H34) are all liquids at room temperature. The viscosity of the alkane liquids also changes with chain length; the shorter the chain the more runny and the more easily it flows, that is it is less viscous. The longer the carbon chain the more viscous (thicker) the liquid alkane becomes.
Which substance is the most viscous?
Which alkane has the largest molecules?
A volatile substance is one that evaporates easily. The smaller the alkane molecule the lower its boiling point tends to be. Small molecules will also have less intermolecular bonding, so it will require less energy to remove particles from the liquid state to the gaseous state. These small molecules are therefore more volatile and evaporate more easily. This is outlined in the image below:
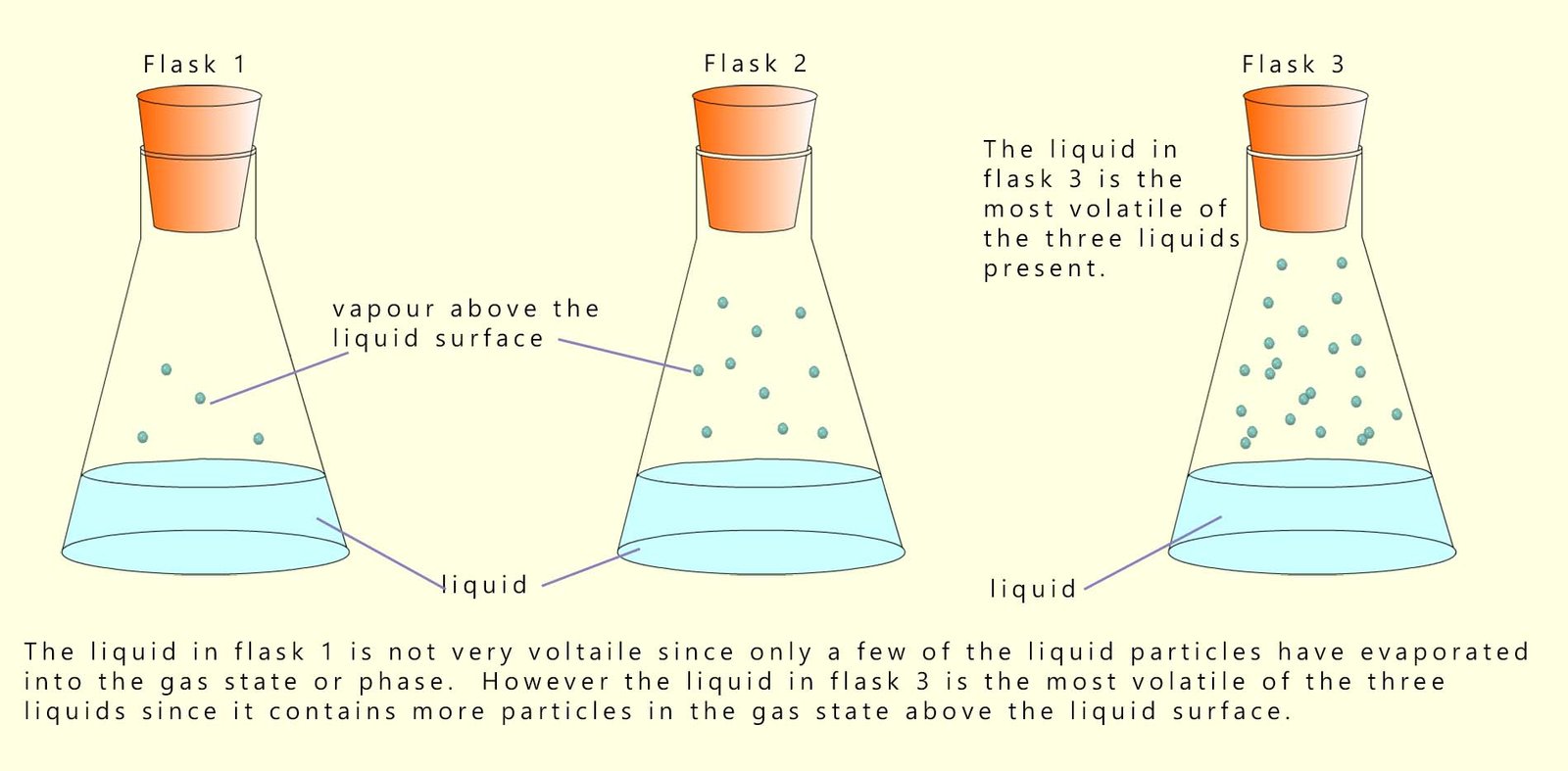
Alkanes which are liquids at room temperature and which contain small molecules will be runny and not particularly viscous and they will be volatile, so they will evaporate easily. As the size of the alkane molecules increases there is more intermolecular bonding between different molecules, this increases their viscosity and decreases their volatility.
 Alkanes are used mainly as fuels. Obviously if an alkane is going to be used as a fuel it must be flammable.
But just how flammable are alkanes? Consider the following example:
Alkanes are used mainly as fuels. Obviously if an alkane is going to be used as a fuel it must be flammable.
But just how flammable are alkanes? Consider the following example:
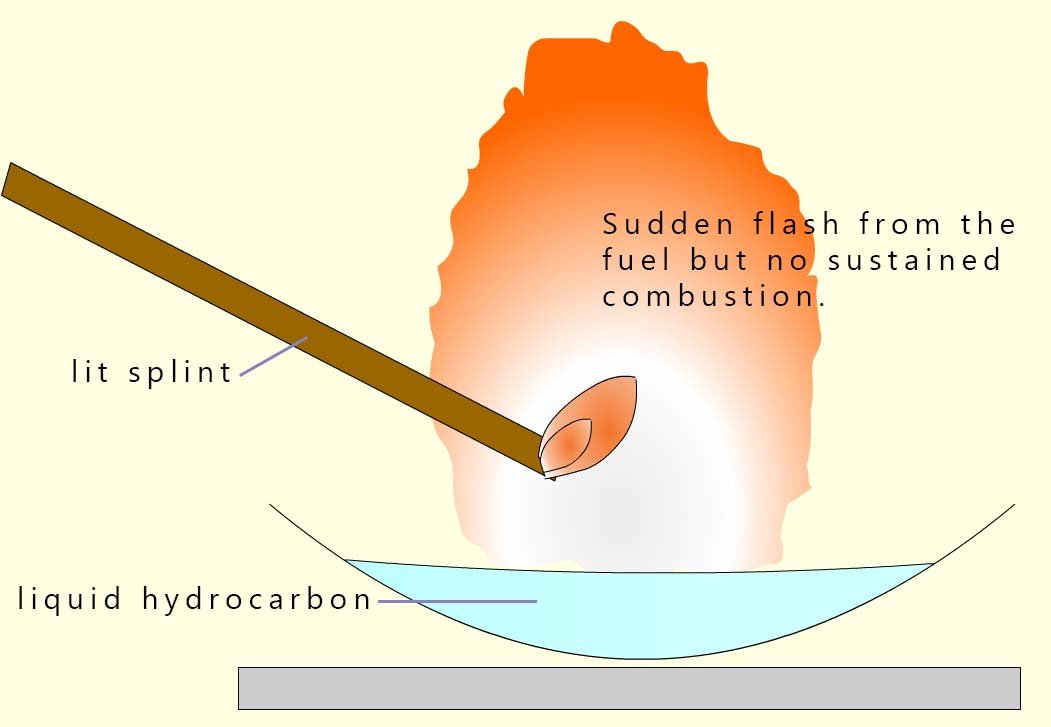
A liquid is placed in an evaporating basin as shown in the image opposite. If the liquid has a low boiling point it will be volatile, that is it will evaporate easily.
If it evaporates easily then above the liquid will be lots of vapour. If the liquid has a high boiling point it will not be volatile and there will be very few vapour particles above the liquid.
It is the amount of particles in the gas phase above the liquid surface that will determine how flammable it is.
When a lit splint is placed above a flammable liquid, it is not the liquid but the vapour above the liquid which burns.
You may hear on TV drama shows such as CSI or fire fighters talking about the flash point of a substance.
The flash point is the point at which there is just enough vapour present above a liquid that when a flame is applied it will flash up, but there is not enough vapour to keep the flame going. The substance will not burn.
However the fire point of a liquid is the point at which there is enough vapour present to sustain combustion and the fuel will continue to burn.
Roughly speaking the fire point is about 10°C higher than the flash point.
The lower the flash point the more flammable the fuel will be.
The table below gives the flash points for several alkane molecules.
| alkane | methane | propane | pentane | heptane | decane |
|---|---|---|---|---|---|
| flash point/°C | -188 | -104 | -49 | -7 | 46 |
From this table you can clearly see that small alkane molecules such as methane have very low flash points and that as the alkane molecules increase in size their flash and fire points will both increase due to the reduced volatility of the larger alkane molecules.
In one sentence, explain why decane is less flammable than pentane.
Match the terms below with their correct definition by simply clicking on the term and then its correct definition. Correct responses will turn the definitions green!
The trend in the flash points and fire points is very clear: the larger the molecule the higher its flash point and hence the less flammable it will be. It is also worth noting that the smaller the hydrocarbon molecule the more cleanly it burns. Large chain hydrocarbons are much more difficult to ignite than smaller chain hydrocarbons and they burn with very dirty, sooty flames. You may also hear people refer to incomplete combustion as dirty combustion because of the soot it produces. You can easily see the soot in a flame by placing a ceramic cup or a clean white evaporating basin in a Bunsen flame. After a few seconds the evaporating basin is black with soot from the dirty Bunsen flame.
Why not use the summary table below to make a set of flashcards to summarise the main points on the physical properties of the alkanes.
| Property | Trend as carbon chain length increases | Why? |
|---|---|---|
| Boiling point 🌡️ | Increases | More intermolecular forces between molecules, so more energy is needed to separate them. |
| Melting point 🧊 | Generally increases | More intermolecular forces in solids, so more energy needed to melt. |
| Viscosity | Increases (gets thicker) | Longer and larger molecules have more intermolecular forces between them and so flow less easily. |
| Volatility 💨 | Decreases | Larger molecules have higher boiling points and stronger intermolecular forces, so they evaporate less easily |
| Flammability 🔥 | Decreases (harder to ignite) | Larger molecules are less volatile, so less vapour above the liquid to catch fire, that is they are less flammable. |
| Flame “cleanliness” | Gets sootier | Larger hydrocarbon molecules need more oxygen to burn completely, so incomplete combustion is more likely |
⚠️ Exam Trap: Many students say the boiling point increases because the covalent bonds get stronger. They do not.
The covalent bonds within the molecule stay the same. What changes is the strength of the intermolecular forces between different molecules.
As relative molecular mass increases, the alkane molecule is larger. Larger molecules have more intermolecular forces, so more energy is needed to separate them. That is why the boiling point increases.
🟦 Viscosity is how easily a liquid flows. A viscous liquid is thick and flows slowly (like honey 🍯). A liquid with low viscosity flows easily (like petrol).
As molecular size increases, intermolecular forces between molecules become stronger. Stronger intermolecular forces make it harder for molecules to slide past each other, so the liquid becomes more viscous.
💨 A volatile substance evaporates easily. Small molecules have weaker intermolecular forces, so they are more volatile.
🔥 The more volatile a liquid is, the more vapour forms above it. It is the vapour that burns, so more volatile liquids are usually more flammable.